Cancer targets and drugs
Most drugs work not only by engaging a single target, but by producing large, complex perturbations of biological systems. In general, the expectation is that the systematic adoption of more rigorous and “systems-level” characterization of chemical entities will help understanding the biology of drug action better and allow the development of improved drugs. It should help the community in rationalizing patient stratification, thus increasing the efficacy of clinical trials and reduce unwanted side effects, but also contribute to the employment of mechanism-based combination therapy with existing drugs. In particular, the GSF lab has a long-standing interest in understanding the molecular wiring of transformed cells of the hematopoietic system, as well as studying the mode of action of targeted agents counteracting leukemia cell proliferation. Starting from BCR-ABL signaling in Chronic myeloid leukemia (CML) to different types of Acute myeloid leukemia (AML) as model system, we are using novel approaches to uncover novel therapeutic avenues and mechanisms of resistance towards targeted therapy.
Metabolic drug library highlights cancer cell vulnerabilities and offer new therapeutic possibilities
Many types of cancer adapt their metabolism to be able to grow and duplicate rapidly and meet the increased bioenergetic and biosynthetic demand. This contributes to the development and progression of cancer. Metabolic reprogramming has been recognized as a hallmark of cancer and may thus represent a vulnerability to be exploited by targeted cancer therapy. The GSF lab in collaboration with the Medical University of Vienna have been working on understanding the dependency of specific functions in human cells on metabolites and nutrients. In our study (Pemovska et al., Nature Communications, 2021), we report the use of a small chemical compound library, called CLIMET (CeMM Library of Metabolic Drugs) targeting a variety of metabolic pathways to identify sensitivities among 15 myeloid leukemia cell lines. The library consists of 243 compounds that influence the metabolism of cells by acting on different branches of the large intricate and widely connected network underlying cellular metabolism. The results highlight specific metabolic “vulnerabilities” of certain leukemia cell types, that may help conceive new therapeutic approaches.
Uncovering relationships between solute carrier transporters and cytotoxic drugs
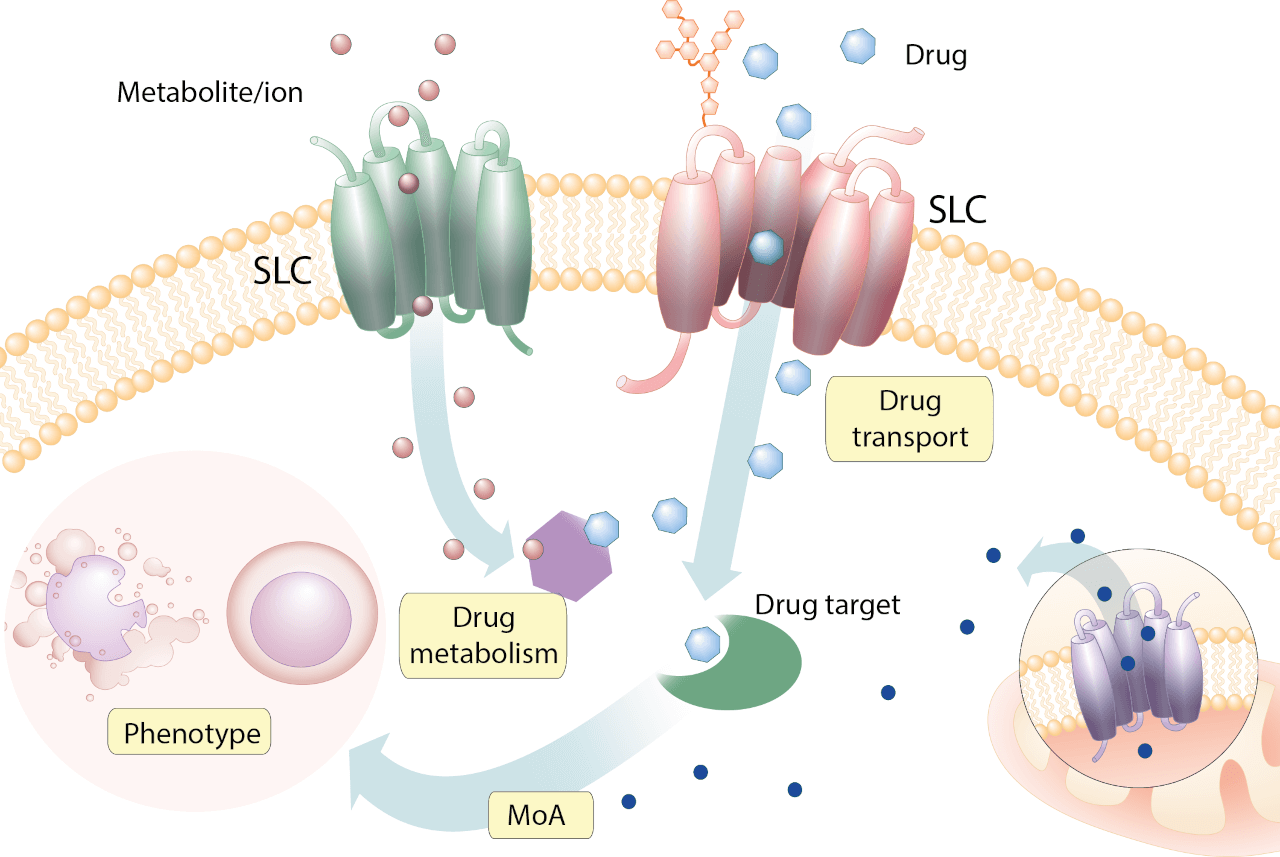
Uncovering how particular drugs enter human cells and how the cell metabolism affects them is key to gaining a better understanding of the side effects and limitations of current drugs and thus developing more effective drug therapies in the future. in what is considered a pioneering study by our laboratory, we found that a particular SLC, SLC35F2 was required for YM155, an experimental cancer drug to enter cells and exert its activity (Winter et al, Nature Chemical Biology, 2014). We recently performed a more systematic investigation on the role of SLCs in determining the activity of a large and diverse set of cytotoxic compounds. Our goal was to survey on the “how often” and “how” SLC transporters would lose or affect the activity of a certain drug. In our study (Girardi et al, Nature Chemical Biology, 2020), the lab built a CRISPR/Cas9-focused library specifically targeting 394 solute carriers and applied it to identify SLCs affecting the activity of a panel of 60 chemically diverse, most of them clinically approved, cytotoxic compounds. We determined that approximately 80% of the screened drug set shows a dependency with at least one solute carrier. This work opens the pathway to further investigations of the genetic determinants of drug activity and especially uptake in human cells. We are convinced that drugs require proteins to enter cells efficiently. Due to the redundancy among transporters and the previous absence of good mammalian genetic ablation technologies have created a false perception on the cell penetrating properties of compounds. As has been argued for a long time by our esteemed colleague Douglas Kell (University of Liverpool) the majority of pharmacology textbooks are wrong in assuming that adherence to the rules set out by Christopher Lipinski a few decades ago is sufficient for drugs to pass membranes by simple diffusion. We concur with Dr. Kell that the excellent Lipinski rules, for what regards permeability, may rather describe the propensity of compounds to engage transporters effectively. Understanding the relationship between the expression of SLCs, cellular/organismal metabolism and nutrition is likely to allow the opening of novel therapeutic avenues in the future.
New mechanism controlling the master cancer regulator uncovered
RAS proteins are central regulators of growth and are found mutated in over 25% of human cancers oncogenesis. Signaling processes around the RAS gene products are also involved in a variety of rare human developmental disorders called the RASopathies. Therefore, every regulator of RAS is poised to be fundamentally important for cancer and a broad variety of human diseases.
Driven by the interest in identifying underlying genetic determinants of drug response in a specific type of cancer of the hematopoietic system, the GSF lab reported on the mechanistic link between RAS and the LZTR1 gene, previously associated with a variety of rare disorders and rare cancers (Bigenzahn et al., Science, 2018). We found that LZTR1, in concert with its co-partner cullin 3, regulates RAS by attaching to it ubiquitin. The modified RAS proteins demonstrate altered localization within the cell and reduced abundance. Mutational defects or inactivation of LZTR1 lead to an increase of RAS dependent pathways causing dysregulation of growth and differentiation. LZTR1 can therefore be considered a breaker of RAS action. Our findings provide a new key regulator of a pathway that is one of the best studied signaling pathways in biology. Our study not only sheds new light and details on the regulation of a central growth-promoting protein, but also offers a molecular explanation for an unusually large number of pathological conditions, ranging from different types of brain and pediatric cancers to developmental pathologies like Noonan syndrome. We are committed to continue to investigate the role of the LZTR1 complex in the proteostatic regulation of the RAS small GTPases and exploit it to derive first-in-class drug candidates.
A New Achilles’ Heel of Blood Cancer
Acute Myeloid Leukemia (AML) is an aggressive form of blood cancer that frequently develops in children. AML is considered a group of leukemias that develop in the bone marrow from progenitors of specialized blood cells, the so-called myeloid cells. Therefore, finding drug targets that affect as many forms of AML as possible is a prime goal for researchers. The GSF lab in collaboration with Florian Grebien’s lab (Ludwig Boltzmann Institute for Cancer Research) and Johannes Zuber’s lab (Institute of Molecular Pathology) were able to identify common, conserved molecular mechanisms that drive oncogenesis in the context of the large number of different MLL-fusion proteins (Skucha, Nature Communications 2018). We identified the methyltransferase SETD2 as a critical effector of MLL-fusion proteins. Using genomic techniques including CRISPR/Cas9 genome editing, we found that SETD2 loss caused induction of DNA-damage and ultimately cell death in the cancer cells. Moreover, SETD2 loss increased the lethal effect of Pinometostat, a drug that is currently in clinical development for treatment of leukemia patients with MLL fusions. These experiments might pave the way for a more effective therapy in the future using a combination of compounds. Pediatric AML is at the center of a project led by PhD student Ben Haladik (also a PhD student of Kaan Boztug at the Children Cancer Research Institute and CeMM adjunct faculty member) aiming at uncovering best possible treatments also for children. For this Ben uses a variety of omics approaches as well as pharmacoscopy, the ex vivo single-cell resolution automated microscopy analysis.
Technology development
The GSF lab has developed or adapted a variety of chemical biology approaches such as chemo-proteomics, genome editing and haploid genetic screens to understand drug action at the molecular level. The laboratory has also developed a small molecule interaction mapping technology using mass spectrometric thermal stability shifts at the proteome-wide level, allowing to investigate the impact of any molecule on intact cells on a global scale (Huber et al., Nature Methods, 2015). Based on these results we have also confirmed that temperature shift works for several-pass membrane transporters, such as SLCs (Hashimoto et al., ACS Chem Biol, 2018).
Drug target identification, drug synergies and mechanisms of drug resistance
Historically, the laboratory has been interested in better characterize the impact of known drugs on cells, using various omics approaches. In the process, one identifies new target candidates for known drugs, previously unknown mechanisms of drug resistance, “effector” genes for the compounds (genes required for the drug to exert its action), mechanisms of synergy between compounds (Winter et al., Nature Chemical Biology, 2012) and, in a few cases, indeed new medical use of existing drugs. By investigating existing drugs with unclear mode of action on rapidly dividing cancer cells and using its arsenal of innovative technologies to uncover their molecular mechanism, the GSF laboratory has identified a new drug target in a poorly characterized pathway involved in the metabolism of oxidized nucleotides (Huber et al., Nature, 2014). Previously, we had identified, for example, that the tyrosine kinase inhibitor dasatinib was active against the tyrosine kinase BTK (Hantschel et al., PNAS, 2007).